Videos: DynaMesh®-HIATUS
Posted: 17. June 2020 | 0 comments

DynaMesh®-HIATUS implantları, basit bir dikiş tekniği kullanılarak hiatoplasti ile yüksek tekrarlama riski bekleniyorsa aksiyal ve/veya paraözofageal hiatal hernilerin cerrahi tedavisi için tasarlanmıştır. Cihazlar, özofageal hiatus bölgesinde diyaframın yumuşak dokusunu kalıcı olarak güçlendirir.
Defined Stability
Constriction of the mesh in the region of the hiatus may reduce the distance between the mesh implant and the oesophagus. DynaMesh®-HIATUS implants have rectangular pores and defined stability with a maximum elongation of only 8% [TR50, TR51].
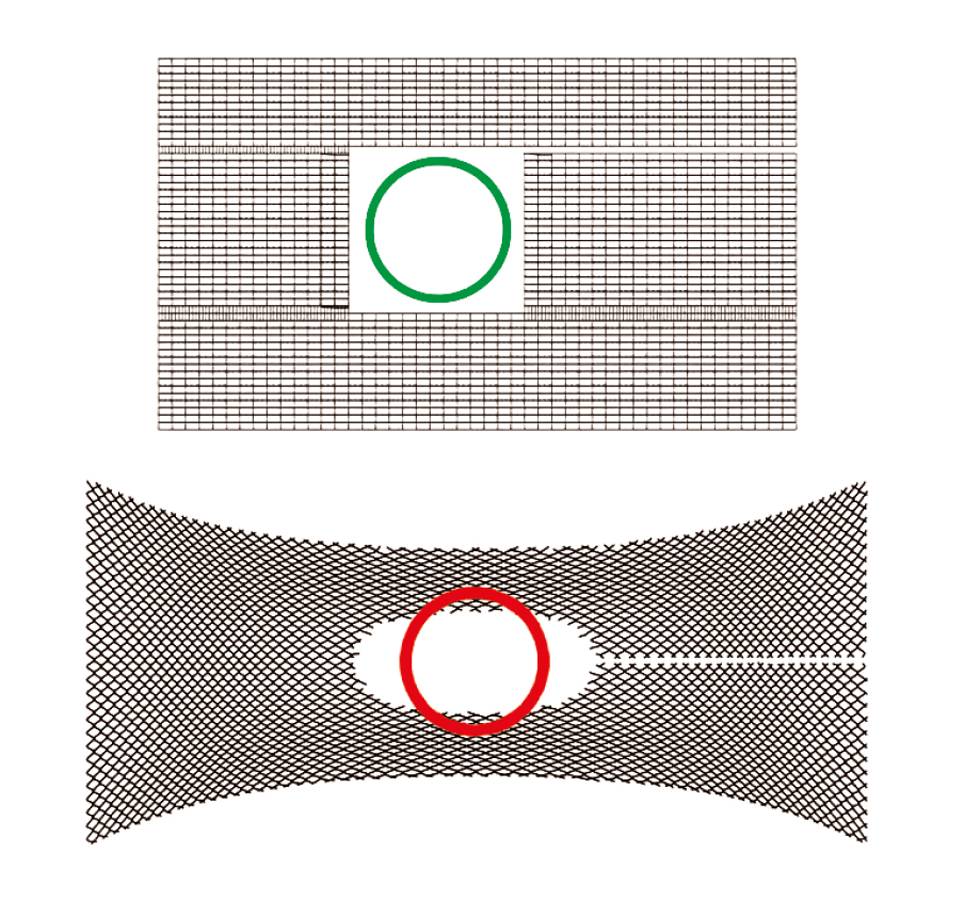
Effective Porosity
All DynaMesh®-HIATUS implants have an effective porosity [TR51]. DynaMesh®-HIATUS implants are made of PVDF. PVDF has good biocompatibility, reducing the foreign body reaction compared to other materials such as polypropylene [1A, 2A, 4A, 68A, 100A, TR1].
DynaMesh®-HIATUS implants show little mesh shrinkage [51].
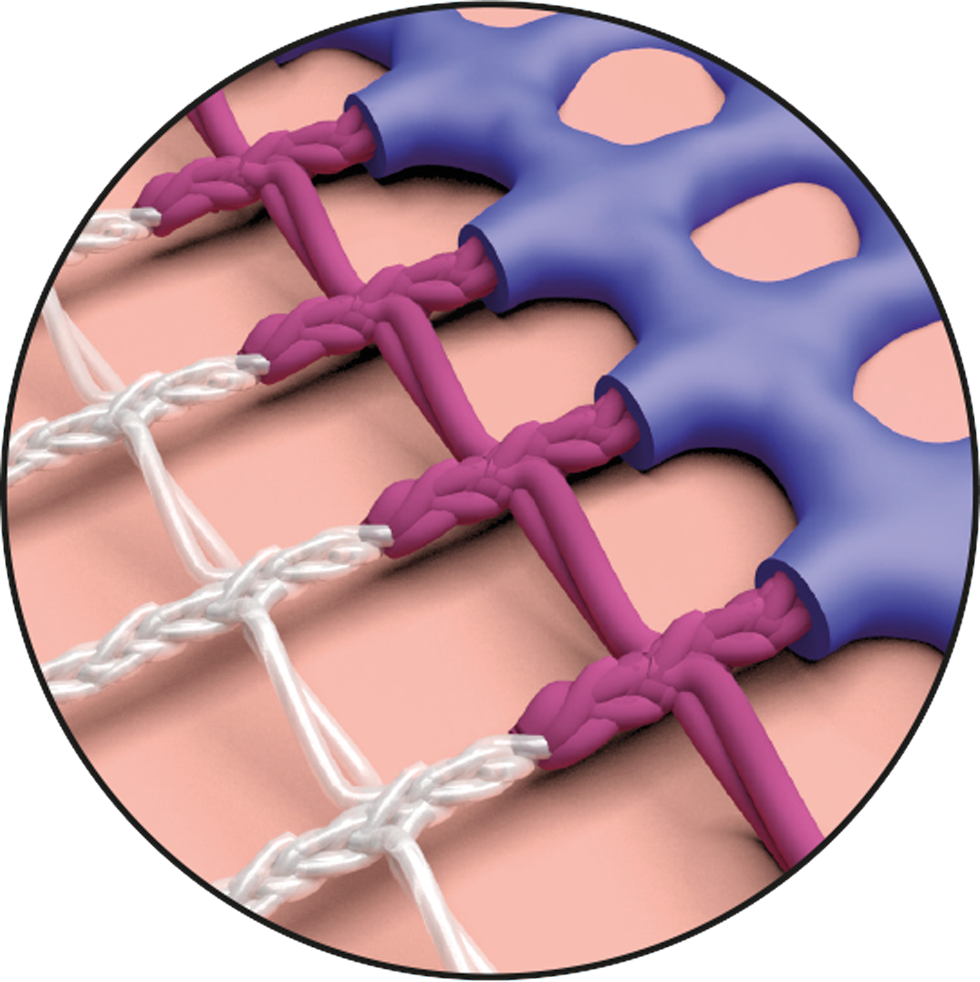
Smooth Warp-Knitted Selvedges
All DynaMesh®-HIATUS implants have smooth warp-knitted
selvedges.
| [#] | Reference “#” (see “References”) |
| [TR#] | Internal test report (see “internal test report references”) |
| Limitations | “A” animal trial, “B” bench test, “VIT” in-vitro trial, “P” published results based on the analysis of human mesh explants, “PB” published results mainly based on bench tests |
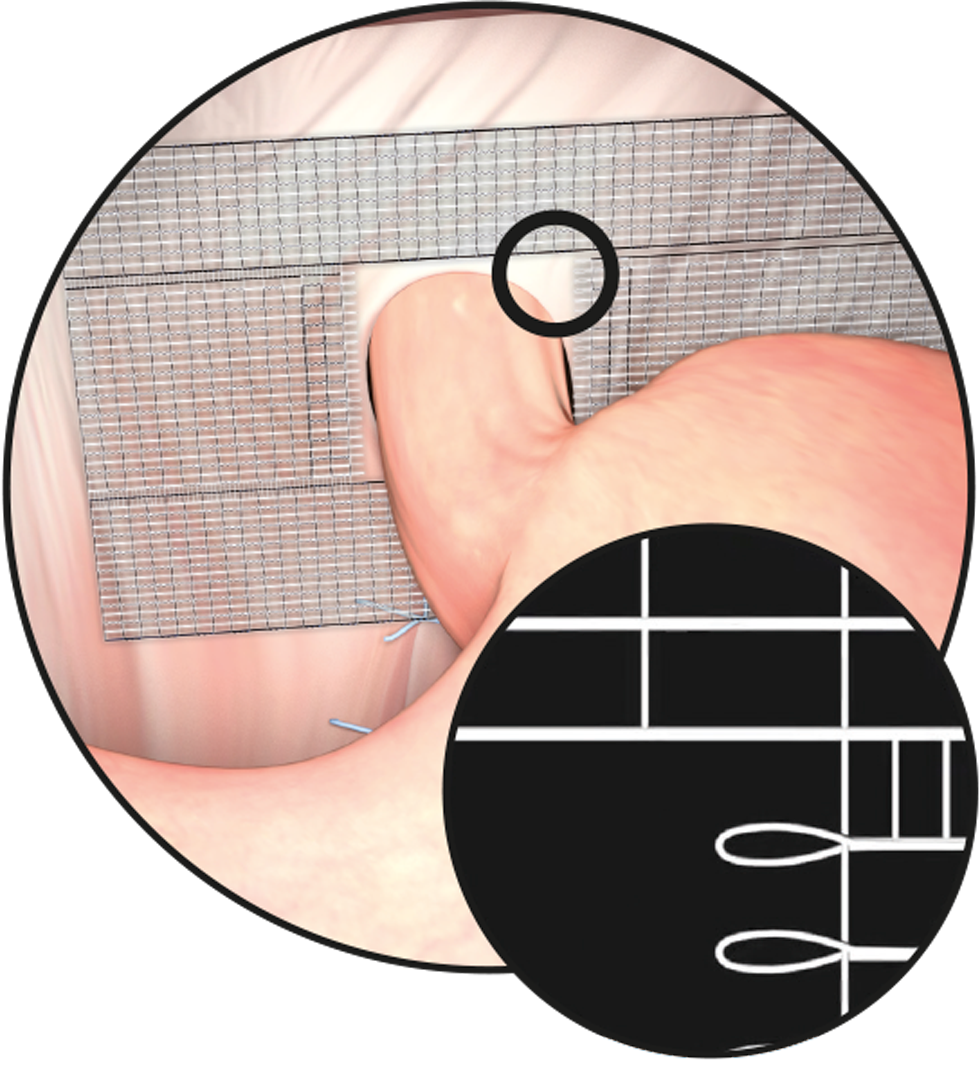
When selecting and cutting the device, ensure that the mesh implant adequately overlaps the hernial orifice on all sides.
| DynaMesh®-HIATUS | 07 cm x 12 cm | PV610712F1/F3 |
| DynaMesh®-HIATUS | 08 cm x 13 cm | PV610813F1/F3 |
| FX = X unit(s)/box (e.g. F3 = 3 unit(s)/box) |
| Product | DynaMesh®-HIATUS |
| Surgical Treatment | Hiatal Hernias |
| Surgical Approach | Minimally Invasive / Open |
| Surgical Technique | Cruroplasty with mesh reinforcement |
| Fixation | Suture* / Tissue Adhesives / Tacks* |
| Defined Stability |
|
| Smooth Warp-Knitted Selvedges |
|
| Visible Technology |
|
| Materials | – Polyvinylidene fluoride (PVDF) (CAS 24937-79-9) > 99% (w/w) – Triiron tetraoxide (CAS 1317-61-9) < 1% (w/w) |
| Polymer (Monofilament) | PVDF |
| Biocompatibility |
|
| Ageing Resistance |
|
| Effective Porosity |
|
| Klinge’s Mesh Classification | Class 1a [102P, TR51] |
| * Traumatic fixation may only be used if injuries to the pericardium, aorta or vena cava can be ruled out with total certainty. |
| Applies to all product sizes | |
| [#] | Reference “#” (see “References”) |
| [TR#] | Internal test report (see “internal test report references”) |
| Limitations | “A” animal trial, “B” bench test, “VIT” in-vitro trial, “P” published results based on the analysis of human mesh explants, “PB” published results mainly based on bench tests |